What Are Three Properties Of Covalent Compounds
Although molecular compounds form crystals they frequently take other forms plus molecular crystals typically are softer than ionic crystals. Start studying Quiz 2.
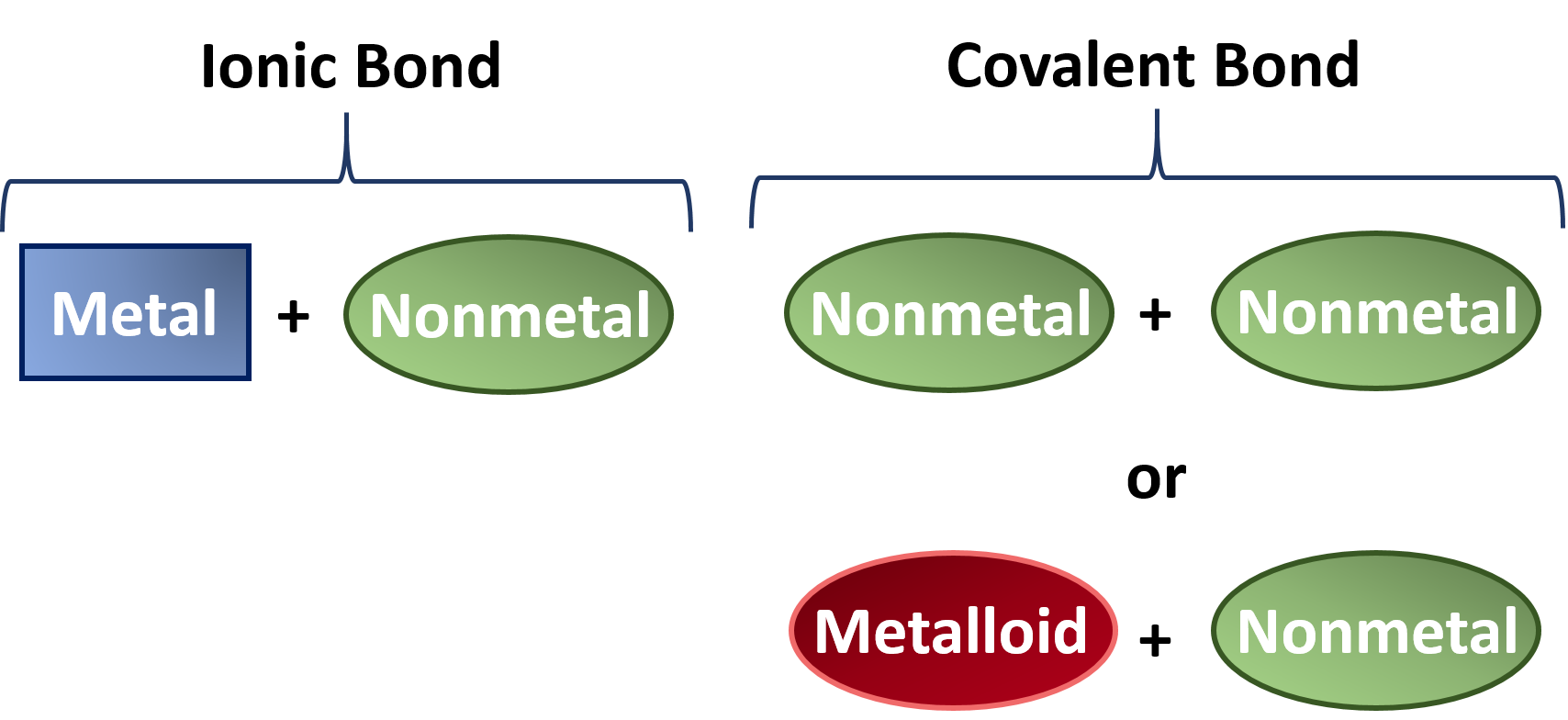
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
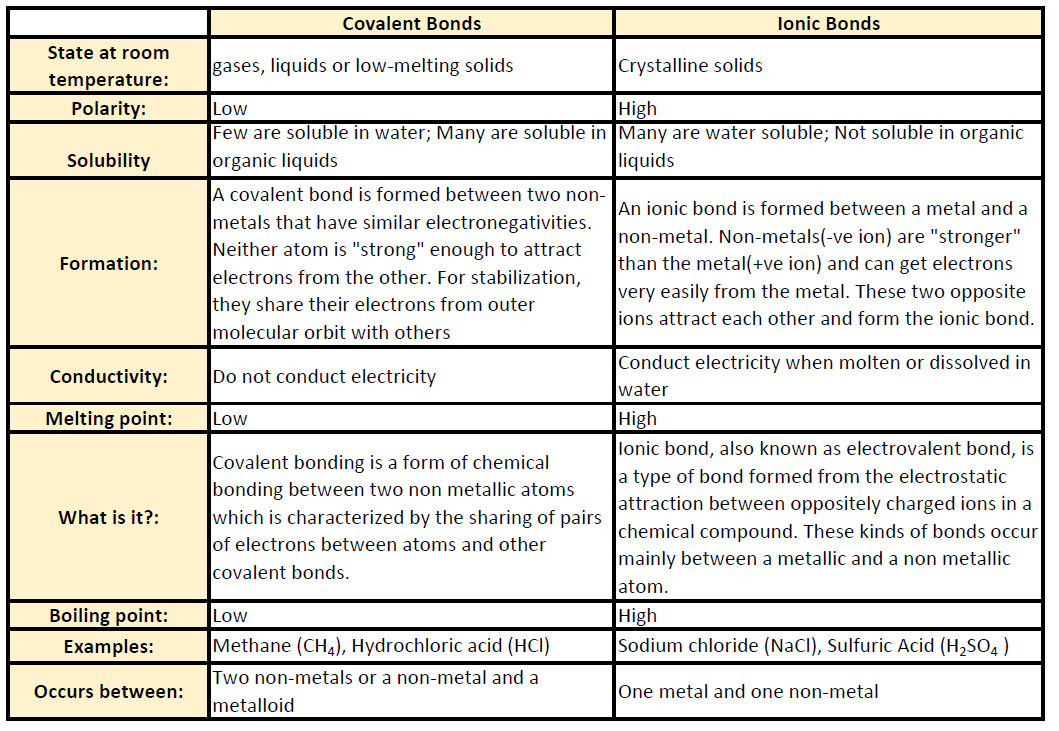
At room temperature and normal atmospheric pressure covalent compounds may exist as a solid a liquid or a gas whereas ionic compounds exist only as solids.
What are three properties of covalent compounds. Ionic compounds form crystals that are composed of oppositely charged ions. At an atomic level an ionic crystal is a regular structure with the cation and anion alternating with each other and forming a three-dimensional structure based. Metal halides can be highly ionic compounds monomeric covalent compounds or polymeric covalent compounds.
This multiple bond ability allows carbon compounds to have a variety of shapes. Covalent compounds generally have low boiling and melting points and are found in all three physical states at room temperature. They are usually obtained through direct combination or through neutralization of a basic metal salt with hydrohalic acid.
The shape of a molecule is determined by the fact that covalent bonds which are composed of shared negatively charged electrons tend to repel one another. Covalent compounds sometimes dissolve well in water eg hydrogen chloride HCl and sometimes do not eg butane C 4 H 10. Unlike ionic compounds with their extended crystal lattices covalent molecules are discrete units with specific three-dimensional shapes.
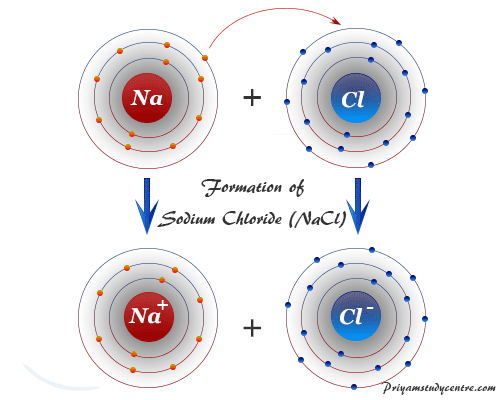
Ionic compounds tend to dissolve in water eg sodium chloride NaCl. Write the formula and predict whether each of the following is principally ionic or covalent by circling I or C. Ionic compounds form crystal lattices rather than amorphous solids.
Lattice energies calculated for ionic compounds are typically much higher than bond dissociation energies measured for covalent bonds. In all carbon compounds carbon forms four bonds. Solid is one of the three main states of matter.
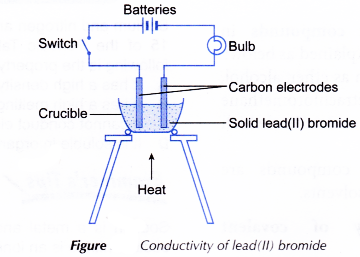
Carbon has the property of forming single double and triple bonds with itself and with other atoms. Network covalent solids and metallic solids. Magnesium chloride crystals sodium sulphate crystals solid leadII bromide diethyl ether hexane cyclohexane distilled water and naphthalene.
Silicon dioxide often called silica is the main compound found in sand. Ions are arranged in a three-dimensional array or crystals. Whereas lattice energies typically fall in the range of 6004000 kJmol some even higher covalent bond dissociation energies are typically between 150400 kJmol for single bonds.
The carbon atom has four electrons in its outermost shell and needs four more. They form crystals. Ionic compounds are generally made up of what kind of elements A metal and a non-metal 2.
These are compounds formed by the reaction of halogens with metals. The types of bonds used by the carbon atom are known as sigma and pi bonds. Learn vocabulary terms and more with flashcards games and other study tools.
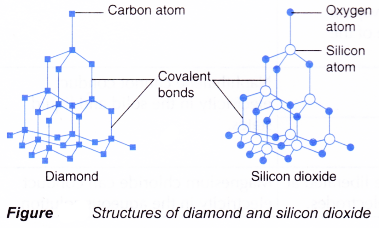
It is an example of a substance with a giant covalent structure. Hence covalent compounds are usually soluble in organic solvents. Properties of ionic and covalent compounds are listed in Table 211.
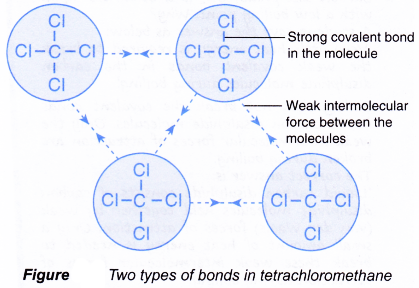
Covalent compounds are generally made up of what kind of elements Two non-metals 3. Here are some differences. Covalent bonds between atoms are quite strong but attractions between moleculescompounds or intermolecular forces can be relatively weak.
44 Shape of Covalent Compounds. Comparison of properties between ionic inorganic and covalent organic. It contains many silicon and oxygen atoms.
To compare the properties of ionic and covalent compounds. Properties of Ionic and Covalent Compounds Experiment. All the atoms.
Covalently bonded substances exist as discrete molecular units. Another example of a nonpolar covalent bond is found in the methane CH 4 molecule. Ionic compounds often dissociate into ions in solution whereas covalently bonded molecules retain their molecular identity in solution.
Nitrogen atoms will form three covalent bonds also called triple covalent between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell. Covalent and ionic compounds can be differentiated easily because of their different physical properties based on the nature of their bonding.
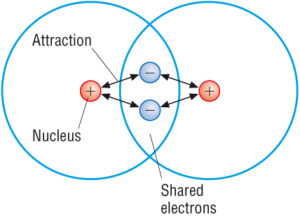
Covalent Compounds Covalent Bond Properties Examples With Videos
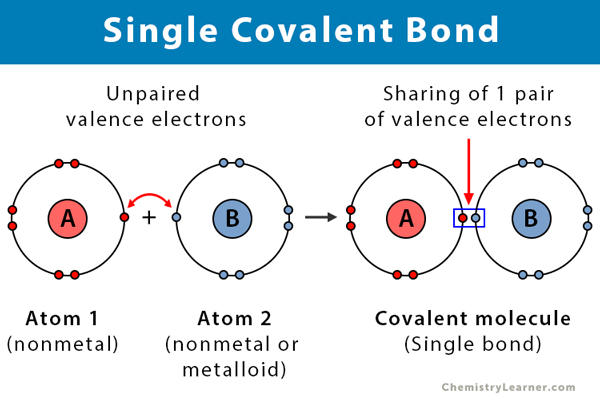
Single Covalent Bond Definition And Examples

Properties Of Covalent Molecules Ppt Download

Properties Of Covalent Molecules Ppt Download

Covalent Bonding In Methane Ammonia Water Properties Of Matter Chemistry Fuseschool Youtube

Covalent Bond Examples Formation Properties What Is A Covalent Bond Video Lesson Transcript Study Com
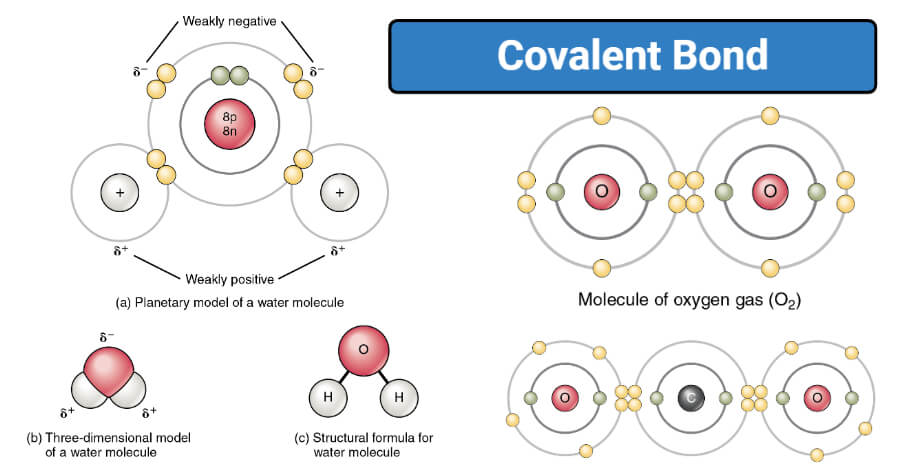
Covalent Bond Definition Properties Types Formation Examples

Covalent Bond An Overview Sciencedirect Topics
Properties Of Ionic And Covalent Compounds A Plus Topper
Properties Of Ionic And Covalent Compounds A Plus Topper
Covalent Bond Types Definition Properties Examples
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Covalent Bond An Overview Sciencedirect Topics
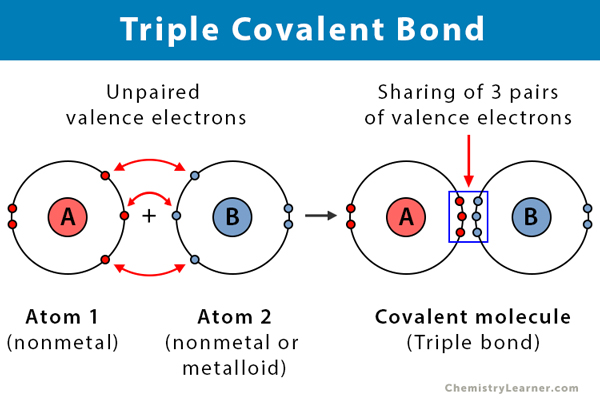
Triple Covalent Bond Definition And Examples

Covalent Compounds Covalent Bond Properties Examples With Videos

Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
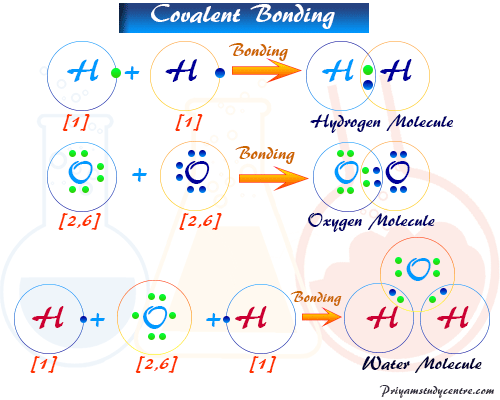
Covalent Bond Types Definition Properties Examples
The Covalent Bond Boundless Chemistry

Properties Of Ionic And Covalent Compounds A Plus Topper
